TRUST STEREQUIP WITH YOUR REPROCESSING
Australia’s first complete external reprocessing solution, you can trust to meet AS 5369.
AS 5369:2023 Reprocessing of reusable medical devices and other devices in health and non-health related facilities
WHY
CHOOSE
US
Strategic Partner
A strategic partnership with Sterequip enables to free up resources, access capital, and repurpose valuable space while maintaining safety and quality.
Efficient Reprocessing
The Sterequip solution provides your health service with external reprocessing of your reusable medical devices with pickup and delivery within next working day.
Securing Capacity
Contracting with Sterequip secures capacity for current and future reprocessing requirements and the ability to meet AS 5369 now and in the future.
Reduced Risk
Sterequip have a team of professionally trained CSSD staff that manage your reprocessing to the manufacturers exacting standard, so each device is reprocessed correctly.
Transparency
Our traceability and automation systems provide full transparency to know exactly where your instruments are and provide security of supply when needed.
Super Centres
Our investment in Reprocessing Super Centres across Australia with state-of-the-art reprocessing equipment and system redundancy to ensure safe, reliable reprocess.
YOUR TRUSTED PARTNER
The Sterequip solution provides your Health Service Organisation (HSO) with external reprocessing of your reusable medical devices with a turnaround of next working day and full tracking and traceability.
We know, smart HSOs only partner with trusted companies that deliver more than operational excellence, with Sterequip you can benefit from:
Income Opportunities
With Sterequip, transform your cost centres into income generating opportunities. Allowing you to remodel valuable CSSD space for activities such as theatres or treatment rooms.
Read more…
Secure Future Capacity
The standard for reprocessing medical devices will change. Secure a complete external CSSD solution or just selected lines or loan equipment so you’re never interrupted by changes to the standard.
Read more…
Manage Multi-Site Requirements
We understand the needs of your individual sites, from preferred pick-up times, instrument type and specialist physician’s personal instruments. Our systems to give you 24 hours visibility.
Read more…
Improving Patient Outcomes
Our team provide the highest quality service to ensure any device is reprocessed correctly every time. Our separate workflows allow us to quarantine a workflow if necessary, giving you greater certainty.
Read more…
STEREQUIP REPROCESSING SUPER CENTRES
Sterequip’s system design is based on the barrier principle, ensuring a clear separation of soiled, clean and sterile zones.
Each zone is separated by a pass-through. The clean system is protected with airlocks that ensure restricted access and positive air pressure.
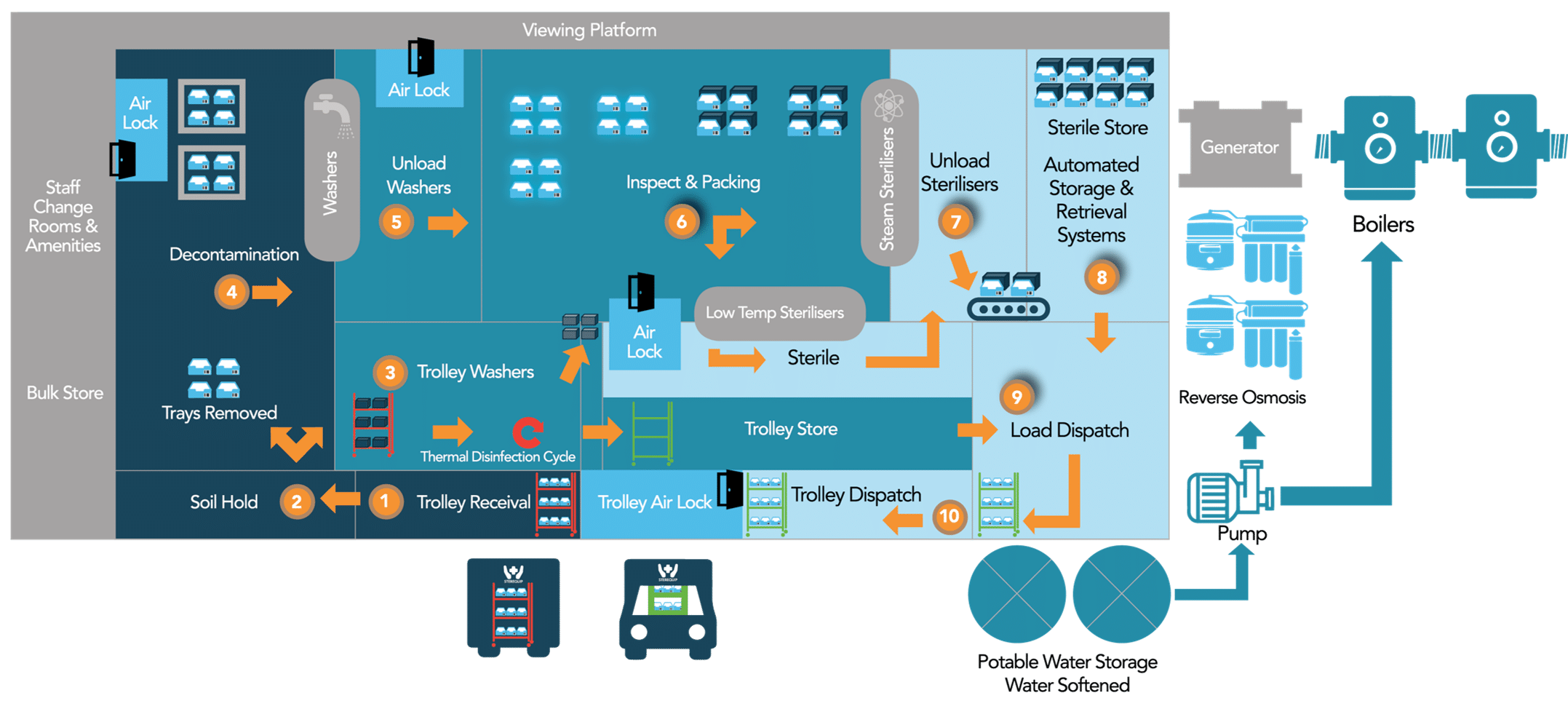
ARRIVAL
1. Trolleys are delivered into the receival dock via an airlock to stop any airborne contamination.
2. Trolleys then move into the Soil Hold then into Decontamination. The decontamination area has an airlock to ensure the material does not leave the area.
WASH
3. Trays are removed from the trolleys and the trolleys go through to the trolley washers and a thermal disinfection cycle of 90 degrees Celsius for 2 minutes.
4. Trays are placed onto washing crates and then passed into one of the 8 washers.
INSPECT
5. Once washed trays move through to the unloading area. Again, the unloading area has an airlock entrance.
6. Trays are then inspected and prepared for sterilisation, either low-temperature sterilisation for heat sensitive items or steam sterilisation. All instruments are returned to the same trays unless repair, maintenance or replacement is needed.
STERILISE & STORE
7. Sterilisation occurs and trays are unloaded and moved on to the Sterile Store or sent through to dispatch for return to the customer.
8. The Sterile Store is fitted with an Automated Storage & Retrieval System.
DISPATCH
9. When required items are retrieved and moved into the Loading and Dispatch area where a technician loads and scans the RMDs into the disinfected trolleys.
10. Trolleys are moved into the Trolley dispatch where they wait until the returning truck is ready for loading. Each day trucks are washed down and UV disinfected. All trucks are temperature-controlled and fitted with humidity sensors.
Income Generating Opportunities & Better Utilisation of Capital
With Sterequip your HSO can utilise valuable CSSD space for higher use activities such as theatres or treatment rooms.
The cost of major infrastructure renovations, new equipment, water and air quality, sterile storage and electronic traceability systems to meet the standard now and in the future will be significant.
Better utilisation of capital into income generating activities allows your HSO to grow providing a competitive advantage for your facilities.

Secure Future Capacity
Two factors affect the growth of your CSSD – the demand for your medical services and changes to AS 5369. Changes to the standard will continue, resulting is additional costs associated with upgrading your CSSD.
The costs escalate especially where space is already limited. The space required to house new utilities, larger equipment and allowance for segregation of the workflow, will impact many HSOs that simply don’t have space in critical areas to meet current or future changes to the standard.
Sterequip can provide either a complete external CSSD solution or manage certain lines or loan equipment to allow your facility to manage growth more effectively.
This means no interruption to your services as you grow or as the standard changes.
Manage Multi-Site Requirements
Sterequip integrates with your tracking and tracing systems to give you 24 hours access to know where your instruments are and manage them as you need.
Our transport solution includes a fleet of temperature controlled and humidity monitored vehicles, designed to segregate used and clean transport trolleys.
With Sterequip we partner with each of your facilities to understand their individual requirements.
Improving Patient Outcomes
When you choose a Sterequip solution, you effectively mitigate risks associated with changes to the standard. Ensuring your instruments are handed and cared for to any future exacting standards.
Our separate workflows allow Sterequip to quarantine a workflow if necessary.
Your facilities have complete visibility on all instruments through our tracking and traceability systems, so you can manage all the data relating to instruments used on your patients.
PROUD
SPONSORS OF


Sterequip Is The Right Choice In Reprocessing
Australian owned and operated, Sterequip is the first in Australia to offer world-class reprocessing super centres to ensure your facilities meet AS 5369 now and in the future.
An alternative to in-house reprocessing, Sterequip is the most reliable, accurate and cost-effective external solution for health services.
Our unrelenting focus on compliance and risk mitigation is achieved with investment into state-of-the-art equipment and systems, and a highly skilled, professional team of specialists in sterilisation and infection control.
Discover how Sterequip can work for you.
COMPLIANCE
Sterequip is committed to providing an AS 5369 compliant reprocessing solution for Health Service Organisations now and for the future changes to the Standard.

